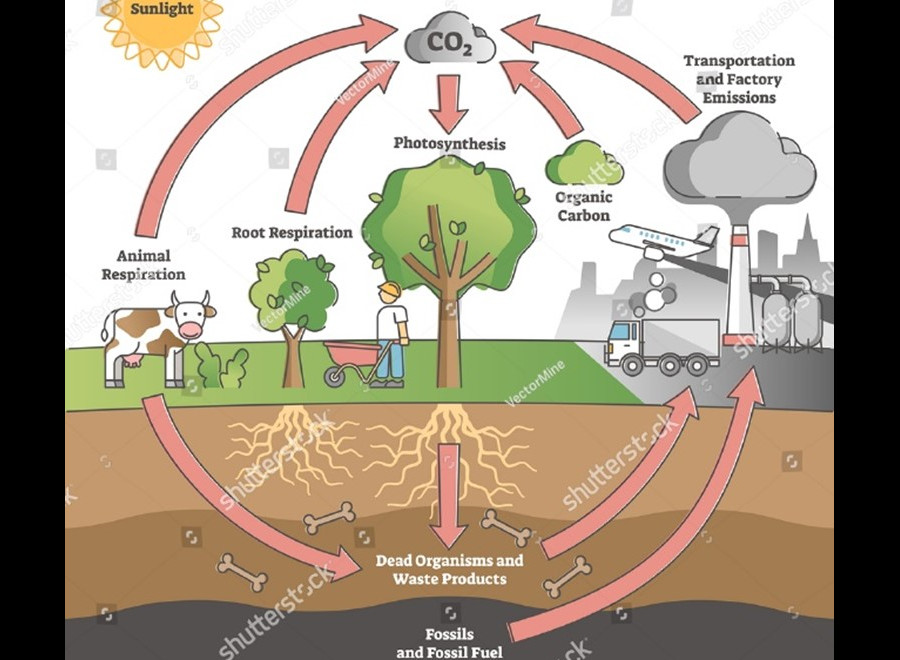
Until 1750, the carbon cycle (CO2) was stable and orderly – the amount of carbon dioxide in the atmosphere was constant because the emitted CO2 was fully absorbed by photosynthesis and the oceans.
Then the coal age began. Moreover, 100 years later, it was the age of oil. Since then, the CO2 produced has not been adequately decomposed in the natural cycle. Due to the industrial revolution, this increase continued to rise sharply from 1945 onwards. Due to deforestation, carbon concentration in the atmosphere has increased by 41% since the coal era and half of that increase was realized in the last 50 years.
The increase in carbon density makes the rainwater acidic and decreases the PH value to 5.
Rainwater that turns into carbonic acid attacks the limestone and concrete stones, causing them to dissolve. In this chemical process, limestone turns into bicarbonate (water-soluble).
CaCO3 + CO2 + H2O -> Ca2+ + 2HCO32-
Limestone + carbon dioxide + water > mixed with calcium + bicarbonate
Sulfur dioxide also makes rainwater acidic. This occurs as a result of some natural gases, coal, oil and waste being released into the air by combustion.
It is now understood why the increase in CO2 and sulfur dioxide poses a danger to concrete and limestone structures and gradually destroys them.
What can we do to prevent this progressive destruction?
Oil; It is used to produce various plastic, resins, acrylates and silicones...
It is common for natural and cement stones to be impregnated with these petroleum products.
Although Oil is the main cause of destruction of natural stones and concrete, it means using oil again for protection and impregnation.
These products only make overfill and do not allow the surface to breathe adequately, causing irreparable damage to the stone they are supposed to protect. They also do not last longer than any applied paint and will decompose very quickly.
They mix with groundwater and are carried into rivers and oceans, where they are absorbed into the food chain as micro plastic particles and re-enter the cycle described above. Therefore, this cannot be the right solution!
How can it be a permanent and 100% natural solution?
Let us examine this with this example. If you take a pebble from the river and break it, you will see that it is wet, damp on the outside, and dry on the inside. Research results, conducted according to the principles of eco-mimetic, have shown that single-celled organisms such as diatom colonies and plankton produce this result. Diatoms consist of free lime and/or liquid silica that crystallizes with CO2, making the stone waterproof. Diatoms thus mineralize the stone.
Jean Pierre Allegre, Technical Manager of ProtecD, developed a mineralizer in his research laboratory that achieves the same result.
Liquid mineralizer, applied to the substrate with a brush, roller or spray bottle, binds CO2 by crystallization without altering the appearance of natural or cement stones.
The mineralizer, which consists of mineral molecules like the stone itself, becomes a whole with the stone and cannot be attacked by the sun, rain or the environment. Protection is absolute.
It protects the concrete structures from pH loss and therefore the metallic parts in the concrete from corrosion.
It makes natural and cement stones more resistant to abrasion by hardening 2 points according to the Mohs scale. It prevents concrete or natural stone from forming dust on its own.
Another positive effect is that it prevents contamination and dirt from penetrating, makes cleaning easier and faster, and preserves the natural colors of stone or concrete.